Particle Size Analysis Lab
The U.S. FDA and other regulatory agencies require particle size testing for nicotine products (e-cigarettes and heated tobacco products [HTP]/heat–not–burn [HNB]). Labstat offers particle analysis testing with the benefit of more than 45 years of industry expertise, customizable testing platforms, and specialized reporting to meet regulatory requirements and support your internal research objectives.
Particle Size Analysis for Regulatory Submissions
The U.S. FDA is denying an overwhelming majority of PMTAs, often due to incomplete submission packets. Everyone in the industry knows how frustrating it is to invest time, money, and resources into a PMTA submission only to receive a denial letter (MDO).
To help your chances of approval, it’s essential to follow the Agency’s guidelines, especially as defined in the FDA’s guidance document, “Premarket Tobacco Product Applications for Electronic Nicotine Delivery Systems: Guidance for Industry”. Particle size distribution testing is a component of the guidance, as highlighted below:
Unit-Only Products
“If the product is manufactured without a solution, you should describe its performance characteristics (e.g., particle size, heating temperature)…” (p. 34)
E-liquids and E-cigarettes Packaged Together
“Additionally, FDA recommends that product testing, such as testing aerosol particle size across the operable range, also be completed using the e-liquid solution and e-cigarette provided in the product package.” (51)
SctnH Scientific Studies
“We recommend that you provide aerosolization properties of each of the ingredients (e.g., constituents, humectants, metals, flavors included), particle size of these ingredients in the product, and deposition of these particles through Inhalation…” (p. 39)
Components/Parts
“Atomizer: Draw resistance (and operable range, if adjustable); E-liquid capacity; and Aerosol particle size across operable range.” (p. 50).
Particle Size Analysis for Research and Development
Our particle size analysis not only assists in modeling the deposition of vapor flavoring droplets but also enables detailed chemical profiling of constituents based on their deposition size. See our presentation at the 76th Tobacco Scientific Research Conference (TSRC) for more information.
Multiple Particle Size Analysis Platforms to Suit Your Needs
Labstat offers multiple platforms for particle size analysis to fit your needs. Our experts can provide guidance on which is right for your project.
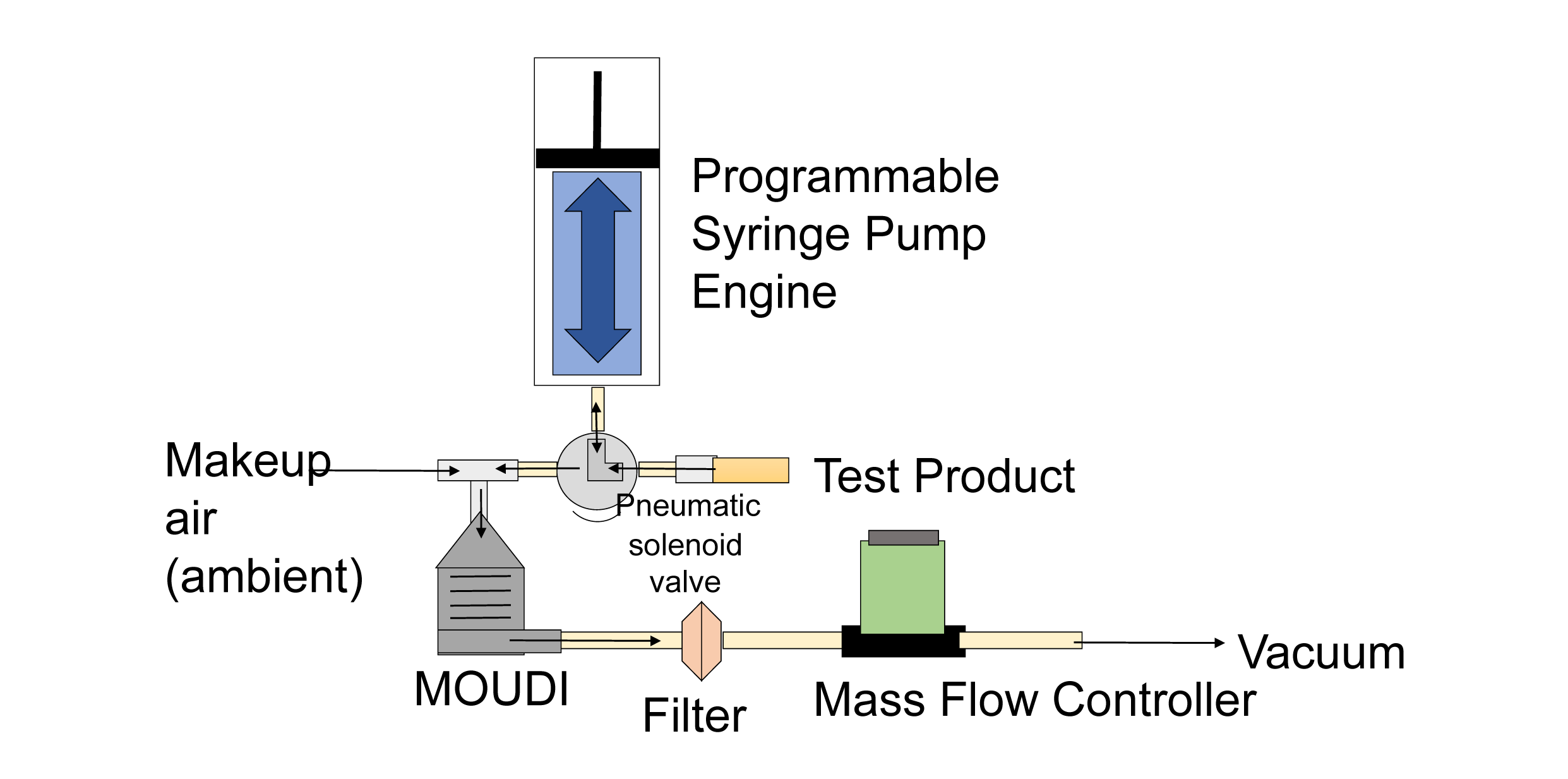
Multi-Orifice Uniform Deposition Impactor (MOUDI)
- Rugged “classic” system suitable for greater stage mass deposition.
- Ideal for chemical post-analysis or 1:1 comparison with common reference data.
Electrical Low-Pressure Impactor (ELPI+)
- Live readout and recording of particle detection enables selective puff processing for end-of-life or profile evolution analysis.
- Ideal for rapid testing of particle size distributions with minimized aerosol manipulation.

Particle Analysis Study Design Options
We can design a particle size analysis study for your product to provide the data you need for research and development as well as your regulatory submissions.
Single-puff block
Alternative substrates for collection
PTFE, metal, polycarbonate
Multi-puff block (“end-of-life”, etc.)
Post-collection analysis
Nicotine, propylene glycol, glycerin, organic acids, carbonyls, glycidol, metals, or custom requests. This work is typically to confirm the detectability and/or distribution of components.
Reporting Options
Services will include technical evaluation and technical customizable reports with the following options:
PM2.5 (as % of distribution)
Mass median aerodynamic diameter (MMAD, µm)
Geometric standard deviation (GSD)
Count median diameter (µm)
MMAD×GSD2 and MMAD÷GSD2 (spans 95% population range)
Particle number conc (#/cm3, ELPI+ only)
Services include technical evaluation and technical report writing as required
Post-Collections Analysis for E-Cigs, HTP, and HNB Options include:
Our Particle Size Testing Lab Provides Data for Regulatory Submissions and Research and Development Projects
Labstat provides the expertise to perform particle size distribution analysis for your regulatory submissions and research and development projects. Complete the short form below to talk to someone today about your next project.

